RESEARCH & DEVELOPMENT
Drive Your Research to the Next Level
OVERVIEW
CGT Global is the ideal partner to accelerate research, clinical trials, and the commercialization of treatments and cures.
Every day, researchers around the world are looking for new treatments and cures for significant diseases. In order to conduct this vital research, they need biospecimens from donors, both healthy and diseased. They use these biospecimens to test applications, discover new drugs, and create new diagnostic and surgical devices.
Having the right high-quality cells is critical to driving research forward. To successfully develop new biological therapeutics for the treatment of disease, researchers need access to a diverse population of donors. They need well-established and consistent protocols for the collection and preparation of human cells, to ensure cell viability and integrity.
With three nationwide CGT Clinics, the CGT Global donor pool is one of the largest HLA-typed and demographically diverse in the industry, with readily available donors that suit your drug discovery and development criteria need well-established and consistent protocols for the collection and preparation of human cells, to ensure cell viability and integrity.
FEATURED PRODUCTS
We have several products that will help accelerate your research applications in humanized mice, toxicology screening, iPSC development, biomarker discovery, cell therapy process development, regenerative medicine research, diagnosis of autoimmune diseases, and side by side comparison studies.
- Human Primary Cells (PBMCs, T-cells, NK-cells and B-cells)
- Cord Blood
- Mobilized Leukopaks
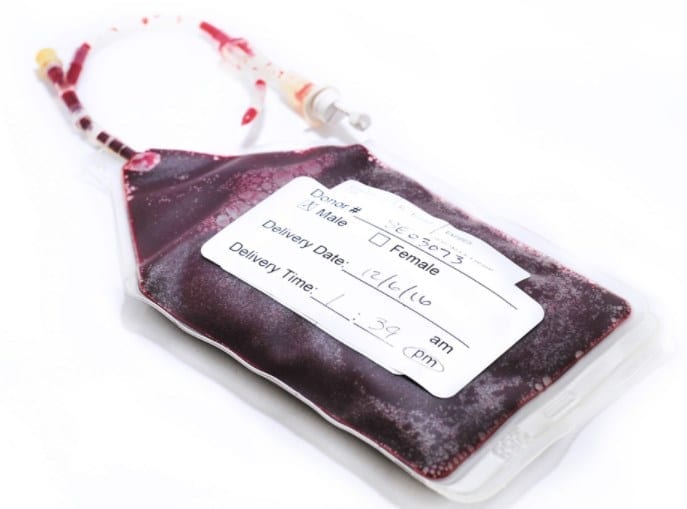
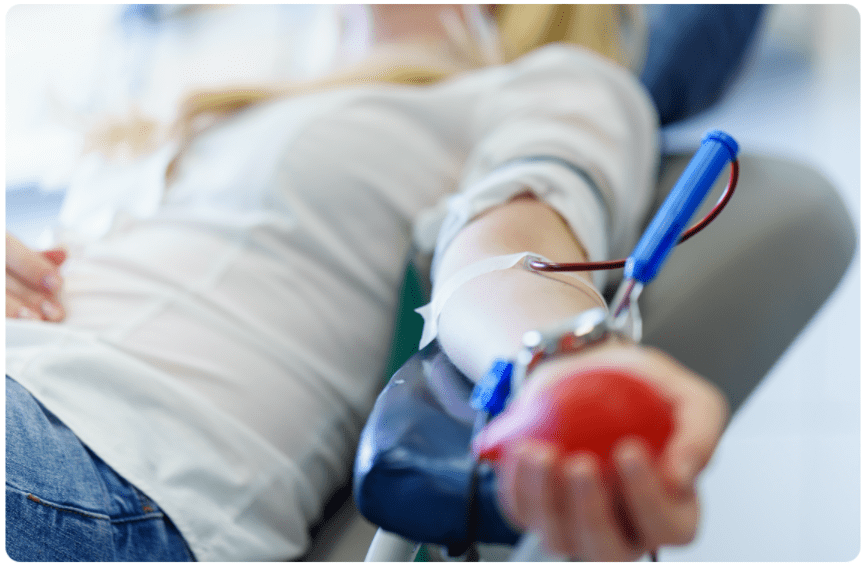
SOLUTIONS
The Right Donor
With three nationwide CGT Clinics, our donor pool is one of the largest HLA-typed and characterized in the industry, with readily available donors that suit your cell therapy criteria. The donor selection process follows strict standard operating procedures and IRB-approved protocols to ensure safe and adequate cell collections.
Quality Products
At CGT Global, we take the necessary steps to ensure regulatory compliance in collecting and producing high-quality leukapheresis products (non-mobilized and mobilized) and primary cells. Each collection and laboratory facility follows a quality system that encompasses Good Manufacturing Practice (GMP) guidelines, industry best practices, and strict quality assurance (QA) management systems to ensure the delivery of consistent, high-quality products.
End to End Process
We provide an end-to-end service whereby cells are collected, processed, isolated, and cryopreserved or shipped out fresh immediately after collection from one of our CGT Clinics. Whether you are requesting tissue or primary cells, fresh or frozen, our staff works with you to schedule a day for a collection that suits your schedule. Additionally, we offer a variety of delivery options, including same-day delivery, depending on your location.
Custom Products
We provide an end-to-end service whereby cells are collected, processed, isolated, and cryopreserved or shipped out fresh immediately after collection from one of our CGT Clinics. Whether you are requesting tissue or primary cells, fresh or frozen, our staff works with you to schedule a day for a collection that suits your schedule. Additionally, we offer a variety of delivery options, including same-day delivery, depending on your location.

