Question about a product? Contact one of our experts. Learn More >
Leukopaks for Cell and Gene Therapy Research and Manufacturing
CGT Global provides high quality human leukopaks to support immune cell research, translational development, and clinical manufacturing across the cell and gene therapy landscape. Leukopaks are collected through leukapheresis and contain highly enriched peripheral blood mononuclear cells including T cells, B cells, NK cells, and monocytes.
These collections provide significantly higher immune cell yields compared to whole blood, allowing researchers to isolate large numbers of immune cells from a single donor. This improves experimental reproducibility and reduces donor to donor variability during cell therapy development and manufacturing.
CGT Global operates FDA registered collection clinics and CLIA certified laboratories that support both research grade and GMP compliant leukopak collections. Our nationwide donor network, recallable donor pool, and same day delivery capabilities allow researchers and biotech companies to access consistent starting material for a wide range of applications.
Leukopaks: Fresh | Frozen | Mobilized
CGT Globals' Peripheral Blood Leukopaks provide enriched peripheral blood mononuclear cells (PBMCs) with low granulocyte and red blood cells. This highly enriched PBMC fraction requires minimal downstream processing (i.e. Ficoll) and can be used directly for isolating high yields of immune cells, such as CD4+ T cells, CD8+ T cells, CD19+ B cells, monocytes, and other immune cells. High cell yields allow researchers greater reproducibility of experiments from a single donor (minimizing donor-to-donor variation), a decrease in same cell variability, and scalability of experiments. In addition, up to 12 Leukopaks can be collected from the same donor in a given year, providing an ample amount of PBMCs for large-scale experiments and research studies. CGT Global also offers Disease State Leukopaks for research.
Leukopaks are sent out on the same day or frozen immediately to ensure a highly viable product.
- 1x1010TNCs/full bag
- Highly-enriched PBMCs
- ≥95% viability (fresh)
- HBV, HCV, HIV negative
- IRB approved
Types of Leukopaks Available
CGT Global provides multiple leukopak formats designed to support different stages of research, translational programs, and clinical manufacturing workflows.
Mobilized Leukopaks
- Fresh
- Cryopreserved
GMP Grade Leukopaks
- Fresh
- Cryopreserved
RUO Leukopaks
- Fresh
- Cryopreserved
Diseased State Leukopaks
- Fresh
- Cryopreserved
FAQ's

Our Facilities
Our CLIA-certified clinical processing lab delivers FDA clinical-grade leukopaks with uncompromising quality — backed by validated protocols, advanced cryopreservation, mobilization, and custom cell enrichment.
Feeding into the lab are CGT Global’s FDA-registered collection clinics in Boston, Philadelphia, and Folsom, equipped with Spectra Optia® systems and staffed by expert nurses and scientists. Supporting these sites is our Reno facility, home to 50,000 sq. ft. of state-of-the-art cryogenic storage capacity. With strategic locations, we offer same-day delivery to major biotech hubs and next-day delivery nationwide, sourcing from a diverse, recallable donor pool to meet your exact study needs.
Boston, MA – Advanced Collection Hub in the Heart of Biotech
Located minutes from the world’s leading research institutions, our Boston facility is FDA-registered and CLIA-certified, equipped with state-of-the-art apheresis technology, and staffed by experienced nurses and scientists. Its proximity to the Greater Boston biotech corridor enables same-day delivery to local labs and rapid turnaround for East Coast clients.
Philadelphia, PA – Scalable Clinical & Research Collections
Our Philadelphia site serves as a high-capacity hub for both GMP and research-grade collections. FDA-registered and CLIA-certified, this location is optimized for large-scale donor programs, mobilized collections, and complex custom processing. Centrally positioned, it offers next-day delivery to most U.S. destinations and efficient global shipping connections.
Folsom, CA – West Coast Speed with National Reach
Strategically located to serve West Coast biotech hubs, our Folsom facility delivers fresh collections to California-based researchers the same day, with rapid shipping across the Pacific and to the rest of the U.S. Fully FDA-registered and CLIA-certified, it offers the same stringent quality standards and customizable donor options as our East Coast sites.
Reno, NV – Cryo-Facilities in Strategic Locations
Located in a major transportation and logistics hub, our Reno headquarters offers a 50,000 sq ft freezer with a -80 degree space.





Coming soon




Fresh Leukopaks
A fresh leukopak refers to a leukopak that has been collected and processed without undergoing significant storage or preservation methods. Essentially, it's a leukopak that is used soon after collection, typically within a short period of time.
Fresh leukopaks are often preferred in research and medical applications where the viability and functionality of white blood cells are critical. This is because white blood cells can be sensitive to storage conditions and may experience changes in activity or function over time.
Researchers and clinicians may opt for fresh leukopaks when they require high-quality, active white blood cells for experiments, treatments, or therapies. For example, in cellular immunotherapy, fresh leukopaks containing active immune cells are often used for the isolation, expansion, and manipulation of specific cell populations for therapeutic purposes.
Fresh leukopaks are collected through leukapheresis procedures, and after collection, they may undergo minimal processing steps before being utilized in research or medical applications. This ensures that the white blood cells remain in a highly functional state and suitable for their intended purpose.
Leukopaks are collected from healthy IRB consented donors, tested negative for HBV, HCV, and HIV, using the Spectra Optia® Apheresis System, where cell-rich mononuclear cells are extracted using a continuous flow system directly into a sterile collection bag containing anticoagulant. Up to 12 Leukopak collections can be performed on the same donor in a given year, providing an ample amount of PBMCs for large-scale experiments and research studies. Additionally, 200-300mL of plasma can be collected at the time of the donor draw.
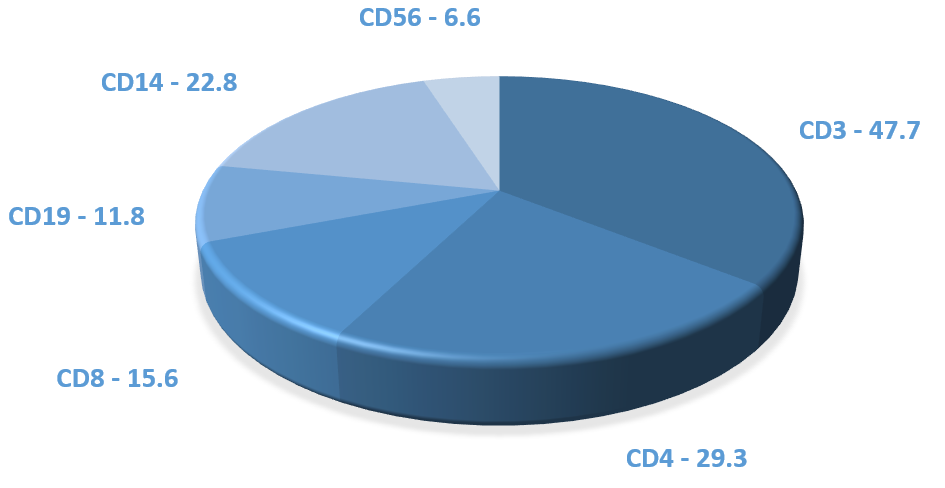
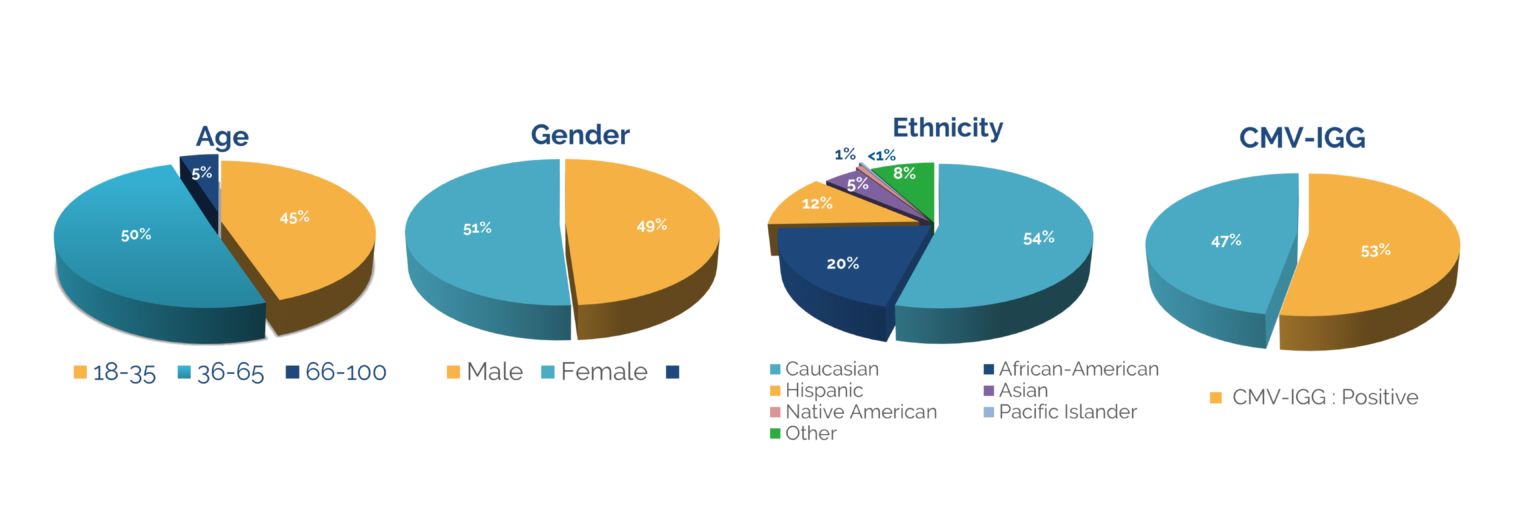
The following are mean percentages of cell populations in a Leukopak:
- T cells – 55%
- Monocytes – 27%
- B cells – 9%
- NK cells – 8%
- CD34+ stem cells – 0.1%
- Granulocytes – 2.4%
We do not guarantee the percentage of cell populations in a Leukopak, as they can vary from donor to donor.
Cells were obtained using Institutional Review Board (IRB) approved consent forms and protocols.
Cryopreserved Leukopaks are collected from IRB consented healthy donors at our FDA-registered CGT Clinics. Using the Spectra Optia® Apheresis System, cell-rich peripheral blood mononuclear cells (PBMCs) are extracted using a continuous flow centrifugation system directly into a sterile collection bag containing an anticoagulant. Besides the PBMCs, which consist of lymphocytes and monocytes, each Leukopak will also contain minimal amounts of granulocytes and red blood cells.
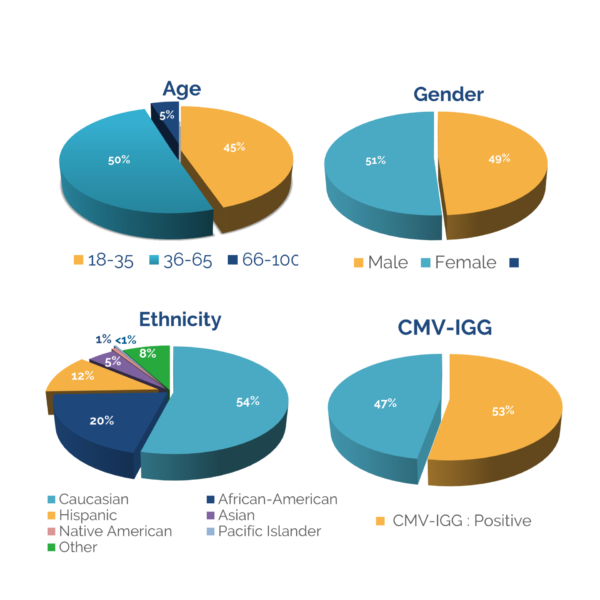
The following are mean percentages of cell populations for a single donor Leukopak prior to cryopreservation:
- T cells – 55%
- Monocytes – 27%
- B cells – 9%
- NK cells – 8%
- CD34+ stem cells – 0.1%
- Granulocytes – 2.4%
We do not guarantee the percentage of cell populations in a Leukopak, as they can vary from donor to donor.

Cryopreserved Leukopaks: Reliable, High-Viability Cells When You Need Them
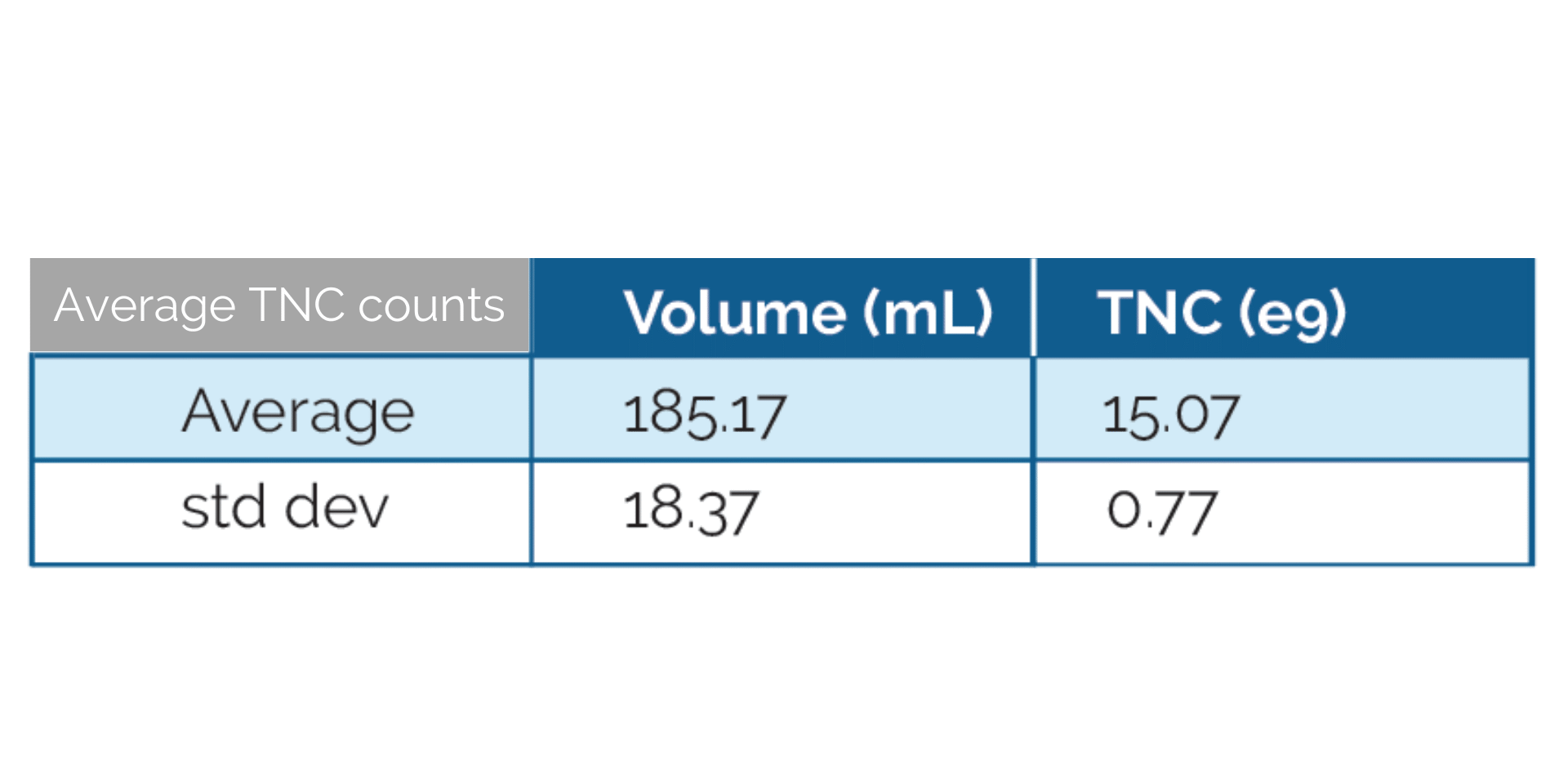
Immediately after collection, Leukopaks are mixed with a cryoprotective medium and frozen using a controlled-rate freezer to preserve maximum cell viability and recovery post-thaw. Prior to cryopreservation, each Leukopak undergoes total nucleated cell (TNC) counts and viability testing to confirm quality and consistency.
To ensure the best results, we highly recommend using our Thawing Protocol (available under the Technical Resources tab). Deviating from this protocol can negatively impact cell viability and recovery rates.
Frozen Leukopak Storage & Shipping
-
Validated LN₂ Cryoshippers: All cryopreserved Leukopaks are shipped globally using LN₂ dry shippers with temperature data loggers, ensuring consistent ultra-cold conditions throughout transit.
-
Stable Long-Term Storage: Leukopaks should be stored in the vapor phase of liquid nitrogen or in -80°C ultra-low freezers until use.
-
Immediate Usability: Thaw and use as needed—flexibility without sacrificing performance.
Why Choose CGT Global’s Cryopreserved Leukopaks?
High Viability & Post-Thaw Performance
Optimized freezing protocols maintain cellular integrity for a wide range of research and clinical applications.
Flexible Scheduling
Avoid timing constraints, thaw on your schedule, not ours.
Global Reach with Guaranteed Quality
Every shipment includes validated cryoshipping, real-time temperature tracking, and rapid delivery from our FDA-registered facilities.
Proven Expertise
Backed by over 15 years of experience in PBMC and immune cell isolation, CGT Global delivers reliability at every stage.
Mobilized leukopaks are collected from donors after administration of a mobilizing agent, most commonly G CSF, to increase circulating immune cells prior to leukapheresis. Compared to non mobilized leukopaks, these collections deliver higher immune cell yields and improved consistency, making them well suited for downstream cell and gene therapy manufacturing.
Mobilized vs Non Mobilized Leukopaks
| Attribute | Mobilized Leukopaks | Non Mobilized Leukopaks |
|---|---|---|
| Immune Cell Yield | Higher and more consistent | Lower and more variable |
| Donor Preparation | Mobilizing agent administered | No mobilization |
| Manufacturing Fit | Supports scale and repeatability | Best for early research |
| Common Programs | CAR T, TCR, NK | Discovery and feasibility |
Mobilized Leukopak Uses
Mobilized leukopaks are selected when higher immune cell yields, reproducibility, and scalability are required.
CAR T Cell Therapy
Mobilized leukopaks support higher T cell availability and more consistent starting material, reducing variability during activation, transduction, and expansion workflows.
TCR Based Cell Therapies
For programs requiring defined T cell populations and tighter manufacturing control, mobilized leukopaks improve process predictability across manufacturing runs.
NK Cell Therapy Programs
Mobilized leukopaks provide increased immune cell availability for NK expansion workflows, particularly for programs planning scale up or repeated manufacturing.

Human Mobilized Peripheral Blood Leukopaks
CGT Global supplies human mobilized peripheral blood leukopaks collected from healthy, IRB consented donors using controlled mobilization and leukapheresis protocols. Mobilization increases circulating immune cells prior to collection, enabling higher cell yields and improved consistency compared to non mobilized leukopaks.
Mobilized peripheral blood leukopaks are commonly used in programs that require reliable starting material for immune cell manufacturing, translational research, and clinical development.
Consistent Cell Yields and Reduced Donor Variability
Mobilized leukopaks support higher and more predictable immune cell yields from a single donor, helping reduce donor to donor variability and improving reproducibility across experiments and manufacturing runs.
For programs advancing toward scale, this consistency can simplify process development, reduce batch failure risk, and support more efficient downstream workflows.
Enriched Immune Cell Populations
Mobilized leukopaks can provide enriched populations of immune cells relevant to cell and gene therapy programs, including:
-
T cells for CAR T and TCR based therapies
-
NK cells for expansion and activation workflows
-
CD34 positive hematopoietic stem and progenitor cells when required
Higher starting cell availability allows teams to optimize isolation, expansion, and manufacturing processes with greater flexibility.
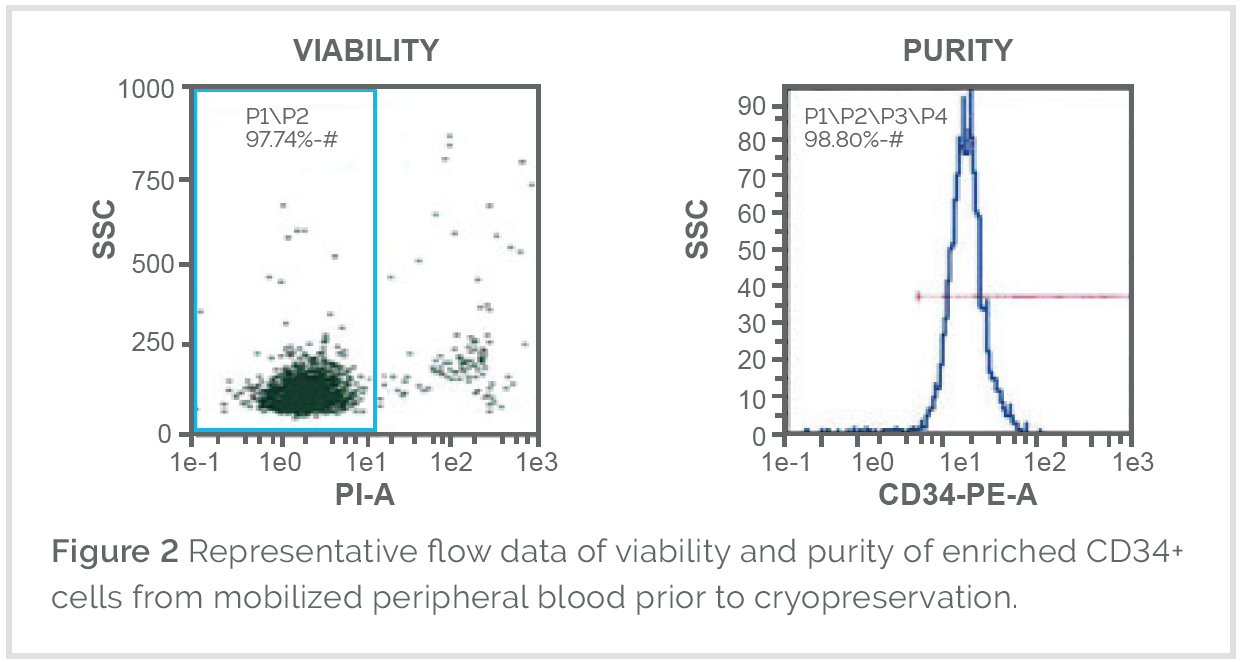
Collection and Quality Characterization
Mobilized leukopaks are collected via leukapheresis by trained technicians using validated apheresis systems. Donors are mobilized prior to collection to support higher immune cell yields and improved consistency.
Following collection, leukopaks undergo quality characterization to support downstream research and manufacturing requirements, including:
-
Flow cytometry verification of immune cell composition
-
Assessment of granulocytes, mononuclear cells, and CD34 positive populations
-
Cell viability confirmation
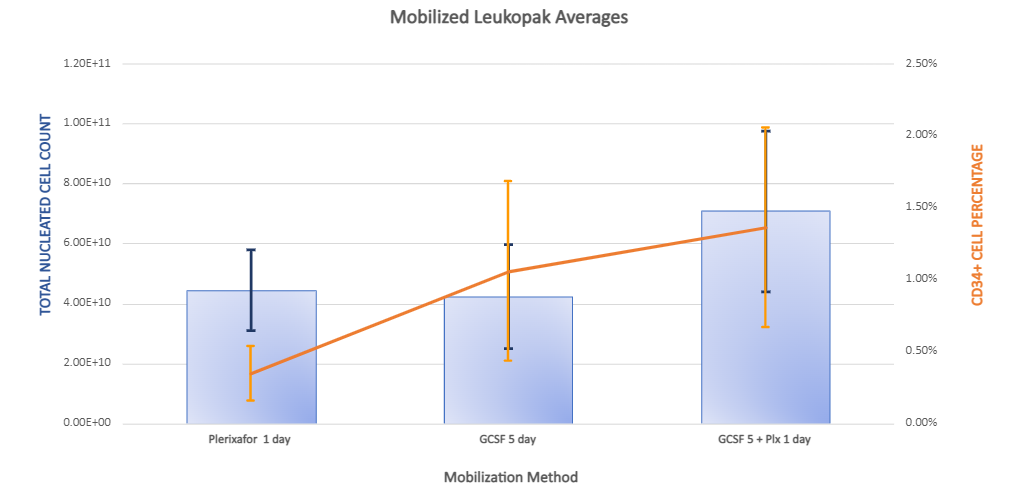
Mobilization increases total nucleated cell yield and CD34 positive cell percentages across collection methods.
Available Formats and Program Support
CGT Global offers mobilized leukopaks in formats designed to support a range of program stages and operational needs.
-
Research grade and GMP grade mobilized leukopaks
-
Fresh and cryopreserved formats
-
Custom donor criteria and collection scheduling
-
Program specific support for research, clinical, and commercial workflows
Related Starting Material and Cell Products
GMP Clinical Leukopaks
Clinical Fresh Leukopak products contain a high concentration of all major lymphocyte populations (T Cells, B Cells, NK Cells, and monocytes) and are GTP/GMP compliant products for cell-based therapy as well as used as the starting material for further manufacturing in allogeneic cell therapies. Our donors follow FDA 21 CFR 1271 guidelines and our productions follow enhanced quality training and are subjected to CMNC protocol in our FDA-registered CGT clinics. Depending on the unique requirements and expectations, CGT Global can customize the donor selection process, infectious disease screening and screening timepoints along with the collection to meet clients needs as well as international regulatory requirements.
What Makes Our GMP Leukopaks Unique?
A GMP (Good Manufacturing Practice) compliant leukopak refers to a leukopak that has been collected, processed, and stored in accordance with strict regulatory guidelines and quality standards outlined by regulatory authorities, such as the Food and Drug Administration (FDA). GMP regulations are designed to ensure the quality, safety, and efficacy of pharmaceuticals, biological products, and medical devices.
For leukopaks to be considered GMP compliant, the entire process, from donor screening to leukapheresis, processing, storage, and distribution, must adhere to GMP principles. This includes implementing standardized procedures, maintaining appropriate facilities and equipment, establishing quality control measures, and documenting all aspects of the collection and handling process.
GMP-Compliant Leukopak Specifications
What does GMP compliance mean for leukopaks?
To be classified as GMP-compliant, leukopaks must be collected, processed, stored, and distributed under strict regulatory oversight, ensuring safety, consistency, and traceability throughout.
Key GMP Elements:
-
-
Donor Screening & Qualification: Following FDA 21 CFR 1271 donor screening and documentation
-
Sterile Aseptic Techniques: For leukapheresis and downstream processing
-
Facility & Equipment Compliance: Collected and processed in FDA-registered facilities
-
Quality Control Protocols: CMNC processing, sterility checks, viability, and cell counts
-
Traceability & Documentation: Every step of the process is documented for audit-readiness
-
Qualified Staff: Trained in cGMP best practices for clinical collections and handling
-
GMP compliant leukopaks are often required for clinical research, investigational drug development, and cell therapy applications intended for use in humans. By adhering to GMP standards, organizations can demonstrate the quality and reliability of leukopaks, ultimately enhancing patient safety and regulatory compliance.

CGT Global Donors
Access to a diverse donor pool is key to the successful development of new therapeutics. With 3 locations nationwide, the CGT Global donor pool is one of the largest HLA-typed and most characterized in the industry. All collection centers are FDA registered, and boast on-site state-of-the-art laboratories, providing an end-to-end service whereby cells are processed, isolated, and cryopreserved or shipped out fresh immediately after collection from a donor.
Choosing a complete partner in research starts with a reliable donor base - from demographic diversity and targeted recruitment to recallability and retention. CGT Global focuses on finding the right donors for a project, with customizable IRB consents and downstream protocols to fit the client's needs. Once a donor is fit to a project, CGT Global works diligently to ensure recallability, with expert donor retention strategies in place.
Donors are an integral part of clinical and preclinical project planning. Knowing how accessible your target donors are, and your ability to reliably recall them can indicate future success and scalability of a project. CGT Global strategically maps out each step in the donor/research process, handling logistics ranging from screening for target genetics, to customizing IRB consents.
In addition to managing donor specifications, CGT Global provides access to a coast-to-coast donor network, with a wide, demographically diverse footprint. Given our far-ranging national reach, CGT Global is able to mitigate supply risks caused by natural disasters, COVID closures, and unforeseen shipping delays.
Our recruitment team seeks out the best donors by engaging with our communities through local businesses, fairs, restaurants, colleges, health and wellness expos, gyms, and donor drives.
- Broad and/or targeted donor recruitment efforts across 5 states
- Collections take place in safe, clean, private collection rooms
- Highly effective donor retention protocols
- Strict guidelines and quality standards - ensuring consistent, compliant, viable, and pure cells with guaranteed counts
- Local same-day delivery within 150 miles
- Recallable and repeat donors
- Customizable donor/patient questionnaire
- Specific donor demographics (age, weight, ethnicity, etc.)
- Donor lifestyle characteristics (BMI, smoker/non-smoker, etc.)
- Medical history (allergies, medications, etc.)
- HLA, RhD, and ABO typing available
- Additional infectious disease screening (CMV, etc.)
- Global reach & delivery
- Good Tissue Practices (GTP)
- All sites FDA Registered
Apheresis
Apheresis is a critical starting point for all research projects, setting the standard for downstream project success through high-quality starting material. CGT Global is a leading provider of apheresis products, with an expansive footprint through our coast-to-coast CGT clinics and unprecedented access to demographically diverse donors. Our expertly trained technicians ensure every draw is GMP compliant, and subsequently shipped out fresh or cryopreserved on-site.
CGT Global collections take place in clean, private, individual collection rooms ensuring a controlled and comfortable experience for donors. Leukopaks are collected in a highly regulated FDA-approved closed system Spectra Optia® Apheresis machine using continuous auditing.
Apheresis collections can be sent to the researcher as a fresh product or sent to the CGT Global lab to be further isolated, or manufactured through custom protocols under the research partner’s or hospital’s regulatory compliance. CGT Global also offers bulk cell isolations from Leukopaks.
Leukapheresis Procedure
Leukopaks are typically collected through a process called leukapheresis that includes the following steps:
- Donor Screening: Potential donors are screened for eligibility based on health criteria to ensure they are suitable for donation. This involves assessing their medical history, current health status, and specific laboratory tests.
- Preparation: Before the leukapheresis procedure, donors receive instructions on fasting or hydration to prepare for the donation.
- Access: The donor is connected to an apheresis machine via one or more intravenous lines. The machine is equipped with sterile tubing and specialized filters.
- Blood Collection: Blood is drawn from the donor and circulated through the apheresis machine. Inside the machine, the blood is separated into its components using centrifugation or filtration techniques in the setting of a leukopak donor center.
- White Blood Cell Collection: The apheresis machine selectively removes the white blood cells from the donor's blood. This process involves separating white blood cells from other blood components based on their physical properties, such as size or density.
- Return of Remaining Blood Components: After the white blood cells have been collected, the rest of the blood, including red blood cells, platelets, and plasma, is returned to the donor's circulation. This helps minimize the impact of the donation on the donor's overall blood volume.
- Collection and Storage: The collected leukopak, containing a concentrated amount of white blood cells, is processed further as needed and then stored under controlled conditions until it is ready for use in research or medical applications.
Overall, leukapheresis allows for the selective collection of white blood cells from a donor while returning the other blood components to the donor's circulation, making it a valuable tool in medical research and therapies requiring a high concentration of white blood cells.

Customization

Customize Your Leukopak
- Fresh or Frozen
- Raw material or isolated cells
- Customized IRB or ICF
- Matched cell types and custom cell counts
- Donor demographics/characteristics
- Shipping, packaging, courier
- Additional infectious disease screening (CMV, etc.)
- Much more....
Additional Services

CGT Global Cell Processing and Isolation
Obtaining a specific primary cell population from human tissue can be time-consuming and costly. If not done quickly and properly, cell viability and recovery can be compromised, creating new challenges to overcome and delays in your research.
To provide high-quality cells to scientists and keep their research moving forward, each CGT Clinic houses an on-site state-of-the-art laboratory, providing an end-to-end service whereby cells are processed, isolated, and cryopreserved or shipped out fresh immediately after collection from a donor. This end-to-end service provides accountability and traceability for each product that leaves our facilities. Additionally, all methods involved in tissue collection, cell processing, cell isolation, and analysis follow strict guidelines and quality standards ensuring consistent, compliant, viable, and pure cells with guaranteed cell counts.
We work with each client to make sure they receive the right primary cells for their project. Therefore, each step of the process is customizable to suit the specific needs of the client, starting with the right donor with specific attributes to isolating and analyzing a specialized cell type.