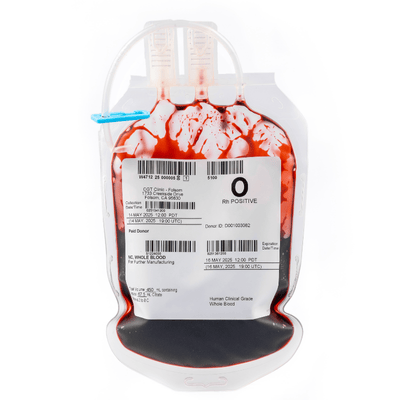
Fresh Mobilized Leukopaks | G-CSF + Plerixafor
Mobilized Leukopaks are collected from healthy IRB consented donors that are injected with a combination of 10µg/kg/day of Granulocyte-Colony Stimulating Factor (G-CSF) and 240µg/kg of Plerixafor the day before collection, which stimulates the bone marrow to produce hematopoietic stem cells and mobilizes them into the bloodstream, prior to first day of collection. Mobilized mononuclear cells are collected by a trained technician using the Spectra Optia® Apheresis System one and two days post injection. After collection, mobilized Leukopaks are checked by flow analysis to verify the percentage of granulocytes, mononuclear cells, CD34+ cells, and cell viability.
- Custom injection regiments with G-CSF and Plerixafor leading to billions of cells per collection
- CD34+ and 7 panel emailed on the day of collection to allow researchers to set up reagents in their lab
- Weekend collections available
- Isolation of CD34+ to save time on isolations and consumables
Since our founding in 2010, CGT Global has pursued our mission to transform healthcare as we accelerate cell and gene therapy research and clinical trials, streamline the commercialization of new treatments, and map the last mile to patient access to these life-changing remedies. By innovating each stage in the cycle; development, commercialization, and delivery, we reduce the overall cost of the care and multiply access points so that millions can receive cutting edge, life-saving gene and cell therapies.

Description
Human mobilized peripheral blood Leukopaks provide a rich source of hematopoietic stem cells as compared to traditional Leukopaks. This allows researchers greater reproducibility of experiments from a single donor (minimize donor-to-donor variation), increase in scalability of experiments from a single product, and a decrease in same cell variability by obtaining high yields of specific cell types.
• 2x10e10 TNCs/1 bag
• Enriched PBMCs
• Enriched CD34+ cells
• Low hematocrit
• Low granulocytes
Cells were obtained using Institutional Review Board (IRB) approved consent forms and protocols.
Custom Orders
We work closely with our customers to provide them with the product they need. For a custom order, please call and speak to one of our sales specialists at 530-303-3828.
Additional information
| Anticoagulant | |
|---|---|
| Format | |
| Grade | |
| Species | |
| Cell and Tissue Source | |
| Disease State | |
| Donor Attributes |
Material Safety Data Sheet
Mobilized Peripheral Blood
Selected publications demonstrating the use of mobilized peripheral blood and mobilized leukopak-derived CD34+ hematopoietic stem and progenitor cells in gene therapy, cell therapy, and transplantation workflows.
Hematopoietic reconstitution dynamics of mobilized peripheral blood-derived stem cells after gene therapy
Nature Communications · 2023
Mobilized peripheral blood-derived hematopoietic stem and progenitor cells were used for gene therapy, showing improved engraftment and recovery compared to traditional sources.
Mobilized Blood · HSPC · Gene Therapy
In vivo CRISPR gene editing in hematopoietic stem cells
Nature Biotechnology · 2022
Mobilized CD34+ hematopoietic stem cells were used in gene editing workflows to evaluate therapeutic genome engineering strategies.
CD34+ Cells · CRISPR · Gene Editing
Ex vivo expansion and functional characterization of hematopoietic stem cells
Stem Cell Research · 2023
Mobilized peripheral blood-derived hematopoietic stem cells were expanded ex vivo and evaluated for functional and therapeutic potential.
HSPC · Expansion · Stem Cell Biology
Optimized lentiviral production and transduction of primary human cells
Molecular Therapy Methods & Clinical Development · 2024
Mobilized CD34+ cells and primary immune cells were transduced using optimized viral production systems for gene and cell therapy applications.
CD34+ Cells · Viral Transduction
Improved workflows for gene modification in hematopoietic stem cells
Molecular Therapy Methods & Clinical Development · 2024
Mobilized hematopoietic stem cells were used to evaluate improved gene modification and cell engineering workflows.
Gene Editing · HSPC
Mobilized peripheral blood stem cells in transplantation and hematologic recovery
Blood · 2023
Mobilized peripheral blood stem cells were used in transplantation workflows to evaluate hematologic recovery and clinical outcomes.
Transplantation · Mobilized Blood